New Article in Journal of Biomolecular NMR
20 November 2023Non-uniform sampling of similar NMR spectra and its application to studies of the interaction between alpha-synuclein and liposomes
Alexandra Shchukina, Thomas C. Schwarz, Michał Nowakowski, Robert Konrat, Krzysztof Kazimierczuk
The accelerated acquisition of multidimensional NMR spectra using sparse non-uniform sampling (NUS) has been widely adopted in recent years. The key concept in NUS is that a major part of the data is omitted during measurement, and then reconstructed using, for example, compressed sensing (CS) methods. CS requires spectra to be compressible, that is, they should contain relatively few “significant” points. The more compressible the spectrum, the fewer experimental NUS points needed in order for it to be accurately reconstructed. In this paper we show that the CS processing of similar spectra can be enhanced by reconstructing only the differences between them. Accurate reconstruction can be obtained at lower sampling levels as the difference is sparser than the spectrum itself. In many situations this method is superior to “conventional” compressed sensing. We exemplify the concept of “difference CS” with one such case—the study of alpha-synuclein binding to liposomes and its dependence on temperature. To obtain information on temperature-dependent transitions between different states, we need to acquire several dozen spectra at various temperatures, with and without the presence of liposomes. Our detailed investigation reveals that changes in the binding modes of the alpha-synuclein ensemble are not only temperature-dependent but also show non-linear behavior in their transitions. Our proposed CS processing approach dramatically reduces the number of NUS points required and thus significantly shortens the experimental time.
New Article in Materials Today Communications
The new Morin – based three-dimensional carbon nanostructures with metal oxides deposits. From synthesis to electro- and photocatalytic applicationsRadon peak-picker based on a neural network
Gisya Abdi, Anna Filip, Krzysztof Kazimierczuk , Tomasz Jaroń, Rafał Jurczakowski, Juan Carlos Colmenares, Andrzej Szczurek

Morin is a natural polyphenolic compound best known for its bioactive properties. Due to its flavonoid structure, Morin may also be considered as a potential precursor of diverse functional materials. Sadly, its poor solubility in water altered its application in this field till now. In this work, by applying simple but effective sol-gel reactions, organic and carbon nanostructures were synthesized and described for the first time. We succeeded in porous materials (BET: 50–600 m2/g) with an original 3D morphology, e.g., hollow hexagonal nanopipes, nanobars, or nanofoams. All received carbons contained metal oxides traces (Co, Zn, Mg, Al), originated from corresponding Lewis acids used as catalysts, and formed after the pyrolysis. The CM-Co samples, having around 7 wt. % of cobalt deposits, were chosen to investigate their suitability for electrochemical conversion of CO2 and photochemical oxidation of benzyl alcohol (BnOH). The CM-Co successfully converted CO2 to CH4 with faradic efficiency (FE) of 12 %, wherein the yield of the reaction was correlated with the applied potential of electrolysis. Furthermore, the CO2 reduction was carried out in ambient conditions. The photochemical oxidation of benzyl alcohol to benzaldehyde yielded 14 % efficiency, while the BnOH oxidation process was realized with a sunlight (UV+VIS) simulator.
New Book Chapter
Chapter 5: The Non-uniform Sampling of 2D NMR Data
Alexandra Shchukina, Paweł Kasprzak, Mateusz Urbańczyk, Krzysztof Kazimierczuk
Conventional acquisition of two-dimensional (2D) NMR signals is based on an equidistant sampling in both time dimensions. The expected signal bandwidth determines the minimum sampling rate; the desired spectral resolution determines the maximum sampling time. Together, these two principles contribute to a minimum number of sampling points required for satisfactory spectrum quality. Additionally, it takes a few seconds for nuclei to relax between the acquisition of consecutive FID signals in a 2D interferogram. These three requirements cause 2D NMR experiments to last even tens of hours, in extreme cases. One of the standard methods to avoid lengthy data collection is to omit a significant proportion of sampling points during acquisition and reconstruct them later using dedicated algorithms. Such an approach to data acquisition is known as non-uniform sampling (NUS). The reconstruction algorithms exploit specific features of the measured signal, usually some form of compressibility. In this chapter, we will discuss the fundamentals of NUS methods including (a) motivation to use NUS in 2D NMR; (b) basic math behind the reconstruction algorithms; (c) commonly used distributions of sampling points; and (d) the use of related approaches in diffusometry, relaxometry, serial experiments and pure-shift NMR.
New Book Chapter
Chapter 10: Fast 2D NMR to Investigate Dynamic Events in Biomolecules
F. Bruno, E. Luchinat, Krzysztof Kazimierczuk, E. Ravera
Interactions among biological macromolecules are often transient in nature, and NMR offers a seat in the front row for observing such events under physiological conditions, thanks to its intrinsic atomic resolution and non-destructive nature. In this chapter, we describe the multidimensional methods that can be applied to monitor biologically relevant events in real time. This includes the combination of rapid pulsing methods and the development of bioreactors for monitoring interactions and reactions in vivo, the use of non-uniform sampling for methods to monitor variations in the state of a protein, and an outlook on the use of paramagnetism to increase the time resolution of NMR acquisition.
New Article in Dalton Transactions
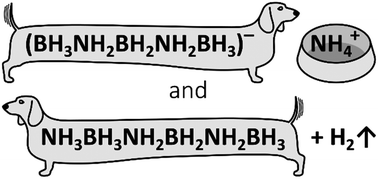
Towards hydrogen-rich ionic (NH4)(BH3NH2BH2NH2BH3) and related molecular NH3BH2NH2BH2NH2BH3
Rafał Owarzany, Tomasz Jaroń, Krzysztof Kazimierczuk, Przemysław J. Malinowski, Wojciech Grochala and Karol J. Fijalkowski
Attempts of the synthesis of ionic (NH4)(BH3NH2BH2NH2BH3) via a metathetical approach resulted in a mixture of the target compound and partly dehydrogenated molecular NH3BH2NH2BH2NH2BH3 product. The mixed specimen was characterised by NMR and vibrational spectroscopies, and the cocrystal structure was analyzed from powder X-ray diffraction data supported by theoretical density functional theory calculations. The compound crystallises in a P21/c unit cell with the lattice parameters of a = 13.401(11) Å, b = 13.196(8) Å, c = 17.828(12) Å, β = 128.83(4)°, V = 2556(3) Å3 and Z = 16. Despite their impressive hydrogen content, similar to ammonia borane, both title compounds release hydrogen substantially polluted with borazine and traces of ammonia and diborane.
New Article in Journal of Physical Chemistry C
Biphasic NMR of Hyperpolarized Suspensions─Real-Time Monitoring of Solute-to-Solid Conversion to Watch Materials Grow
Ertan Turhan, Christopher Pötzl, Waldemar Keil, Mattia Negroni, Karel Kouřil, Benno Meier, Javier Agustin Romero, Krzysztof Kazimierczuk, Ieva Goldberga, Thierry Azaïs, and Dennis Kurzbach

Nuclear magnetic resonance (NMR) spectroscopy is a key method for the determination of molecular structures. Due to its intrinsically high (i.e., atomistic) resolution and versatility, it has found numerous applications for investigating gases, liquids, and solids. However, liquid-state NMR has found little application for suspensions of solid particles as the resonances of such systems are excessively broadened, typically beyond the detection threshold. Herein, we propose a route to overcoming this critical limitation by enhancing the signals of particle suspensions by >3.000-fold using dissolution dynamic nuclear polarization (d-DNP) coupled with rapid solid precipitation. For the proof-of-concept series of experiments, we employed calcium phosphate (CaP) as a model system. By d-DNP, we boosted the signals of phosphate 31P spins before rapid CaP precipitation inside the NMR spectrometer, leading to the inclusion of the hyperpolarized phosphate into CaP-nucleated solid particles within milliseconds. With our approach, within only 1 s of acquisition time, we obtained spectra of biphasic systems, i.e., micrometer-sized dilute solid CaP particles coexisting with their solution-state precursors. Thus, this work is a step toward real-time characterization of the solid–solution equilibrium. Finally, integrating the hyperpolarized data with molecular dynamics simulations and electron microscopy enabled us to shed light on the CaP formation mechanism in atomistic detail.